Can the rate of effusion or diffusion be negative, in accordance with Graham's law? If so, how? - Quora

acid base - What is the relative rate of diffusion of ammonia to hydrogen chloride, both in gaseous states? - Chemistry Stack Exchange
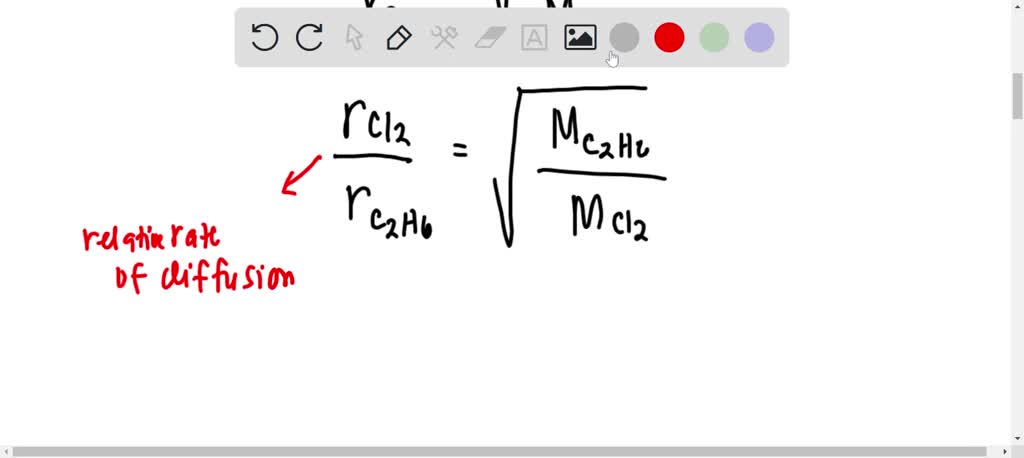
63.Rate of diffusion of gas X is twice that of gas Y if molecular mass of Y is 64 then the molecular mass of X will be

Graph of diffusion constant versus temperature. Solid lines represent... | Download Scientific Diagram
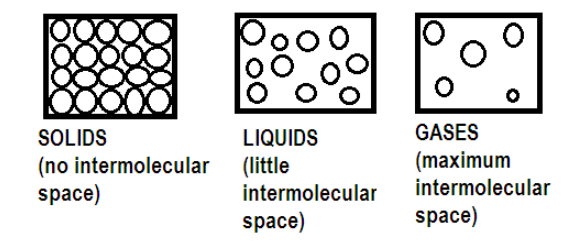
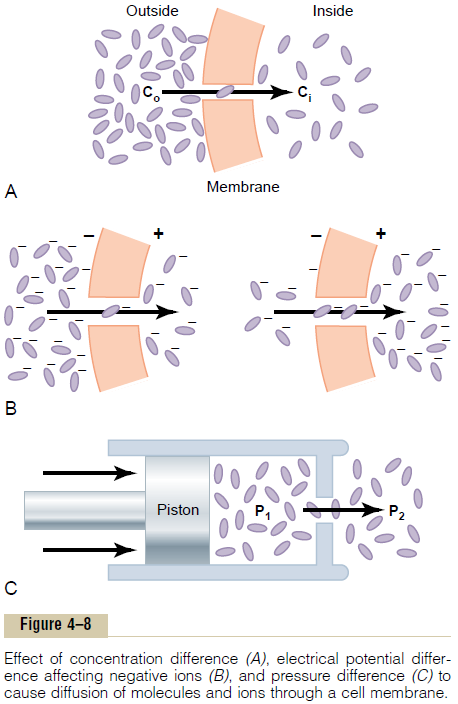
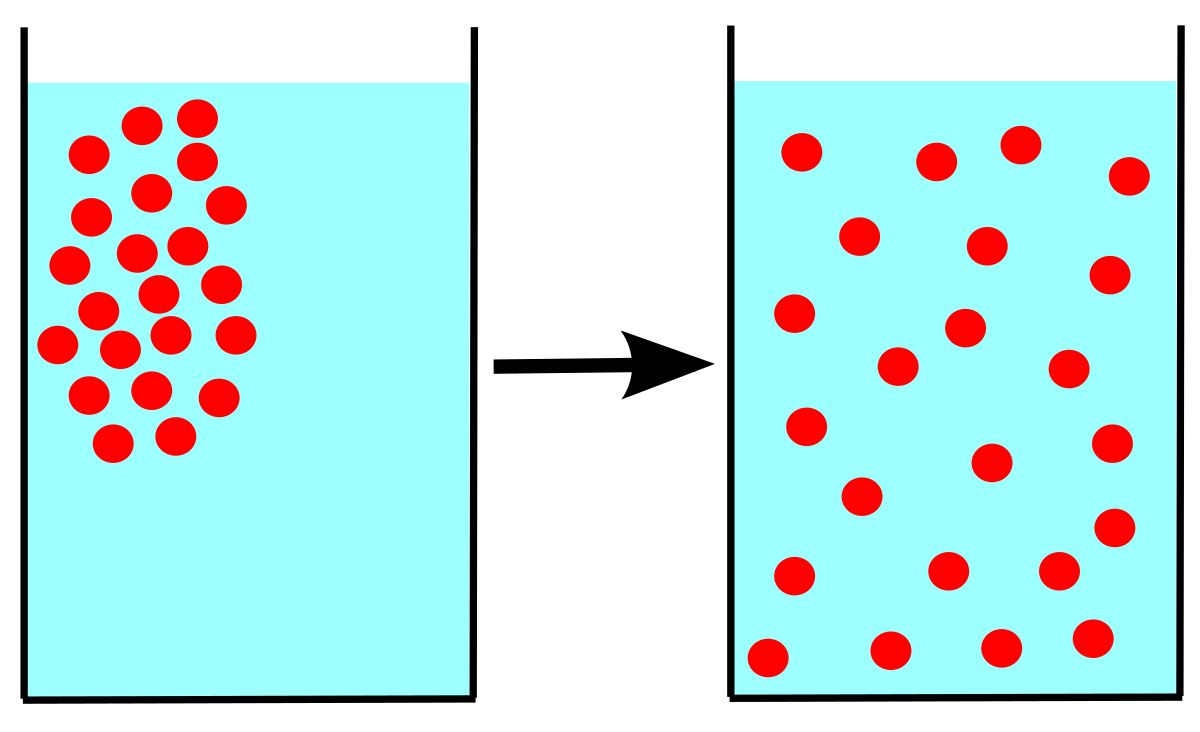
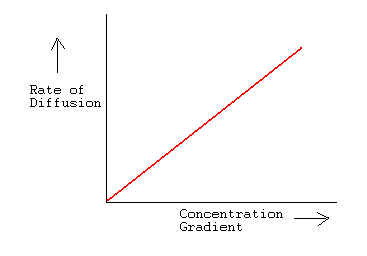
The rate of diffusion of liquids is ______.A) Higher than gases.B) Lower than solids.C) Higher than solids. D) Equal to gases.