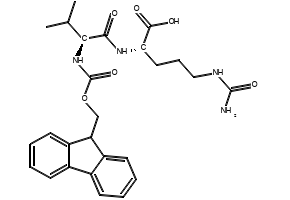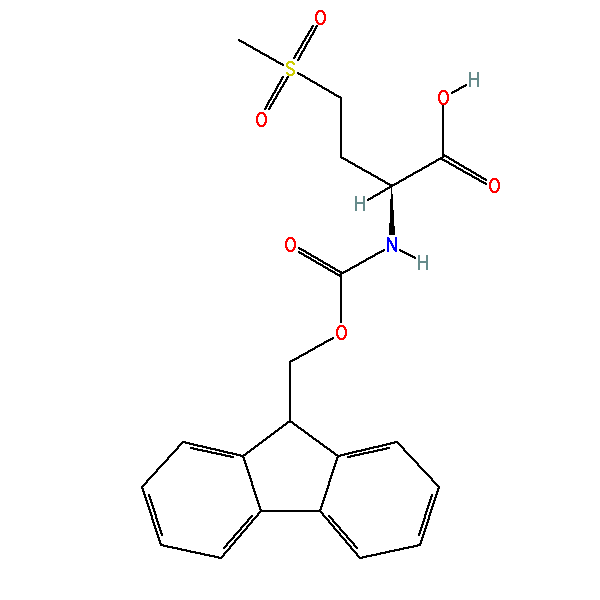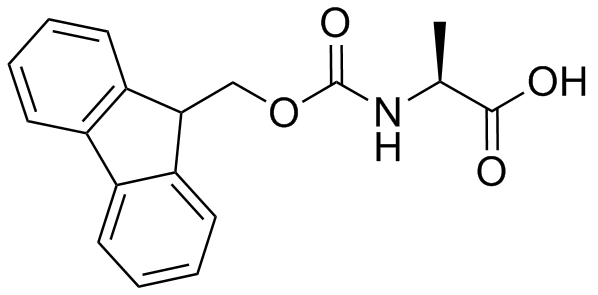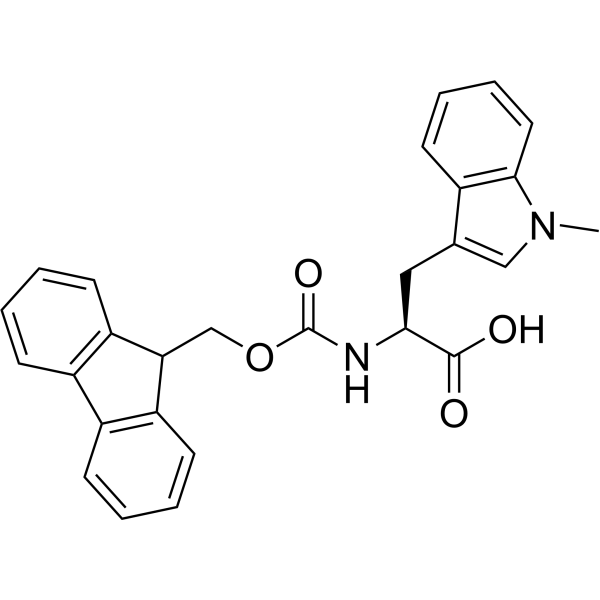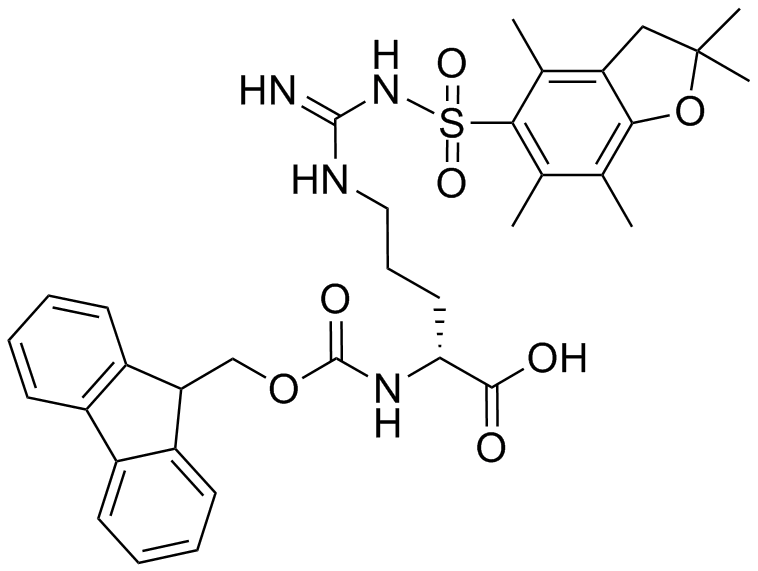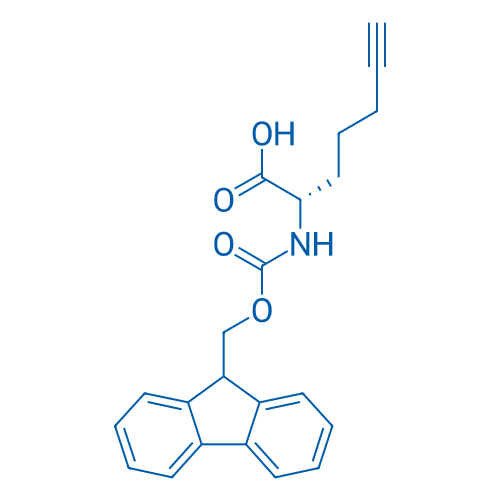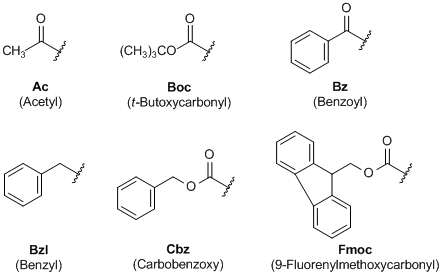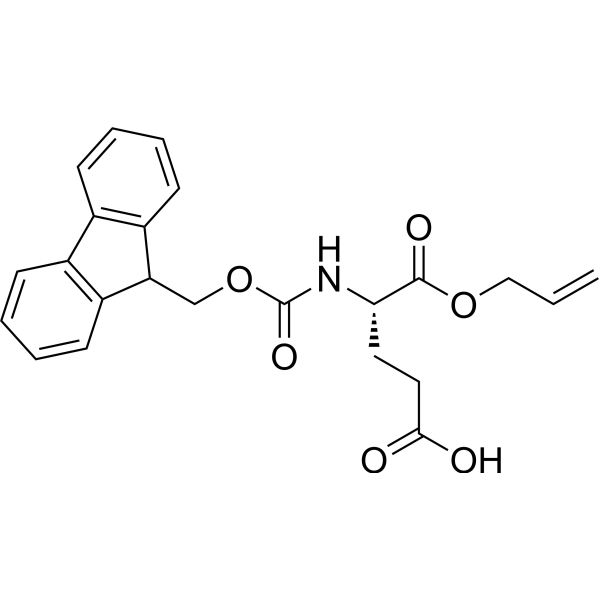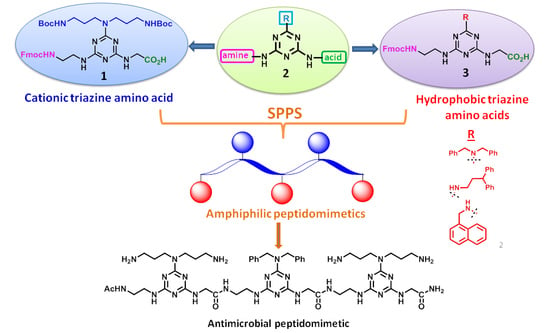
IJMS | Free Full-Text | Synthesis of Fmoc-Triazine Amino Acids and Its Application in the Synthesis of Short Antibacterial Peptidomimetics
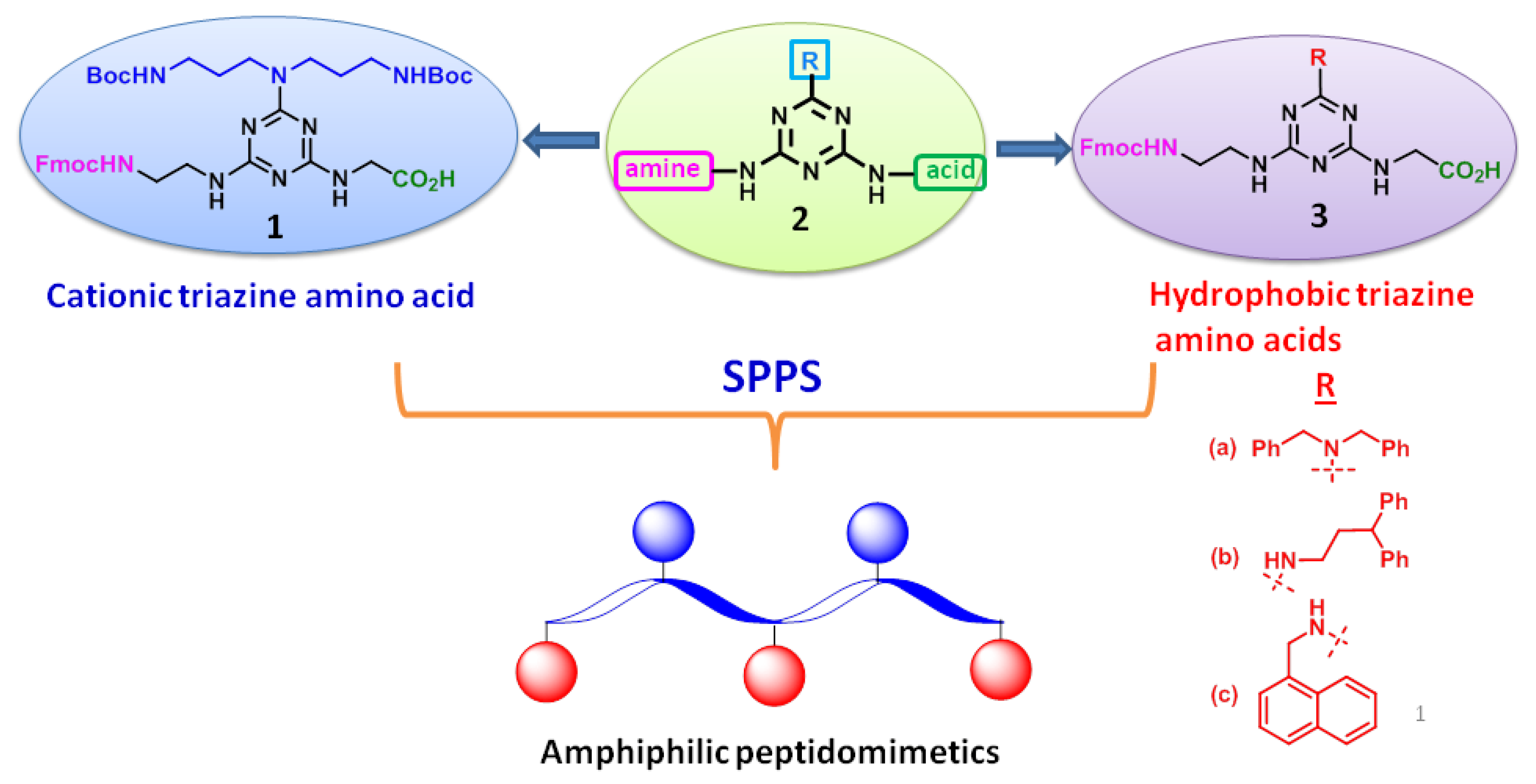
IJMS | Free Full-Text | Synthesis of Fmoc-Triazine Amino Acids and Its Application in the Synthesis of Short Antibacterial Peptidomimetics

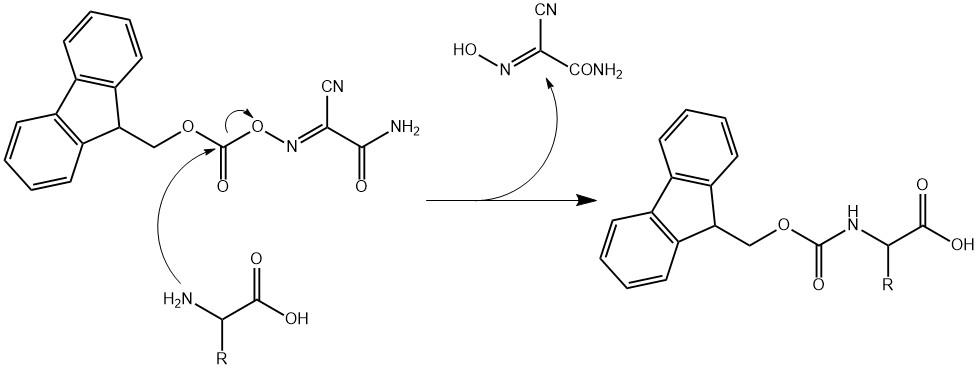
Fmoc-OASUD: A new reagent for the preparation of Fmoc-amino acids free from impurities resulting from Lossen rearrangement - ScienceDirect
Fmoc-L-Proline, 50 g, CAS No. 71989-31-6 | Fluorenylmethylene / Fmoc | Amino acids, protected | Amino Acid Derivatives | Amino Acids and Amino Acid Derivatives | Organic & Bioorganic Chemicals | Chemicals | Carl Roth - International

Aza-Amino Acid Scanning of Secondary Structure Suited for Solid-Phase Peptide Synthesis with Fmoc Chemistry and Aza-Amino Acids with Heteroatomic Side Chains | ACS Combinatorial Science

An Fmoc protecting group can be removed from an amino acid by treatment with the amine base piperidine. Propose a mechanism. | Homework.Study.com
Fmoc-L-Cysteine-(Acetamidomethyl), 5 g, CAS No. 86060-81-3 | Fluorenylmethylene / Fmoc | Amino acids, protected | Amino Acid Derivatives | Amino Acids and Amino Acid Derivatives | Organic & Bioorganic Chemicals | Chemicals | Carl Roth - International